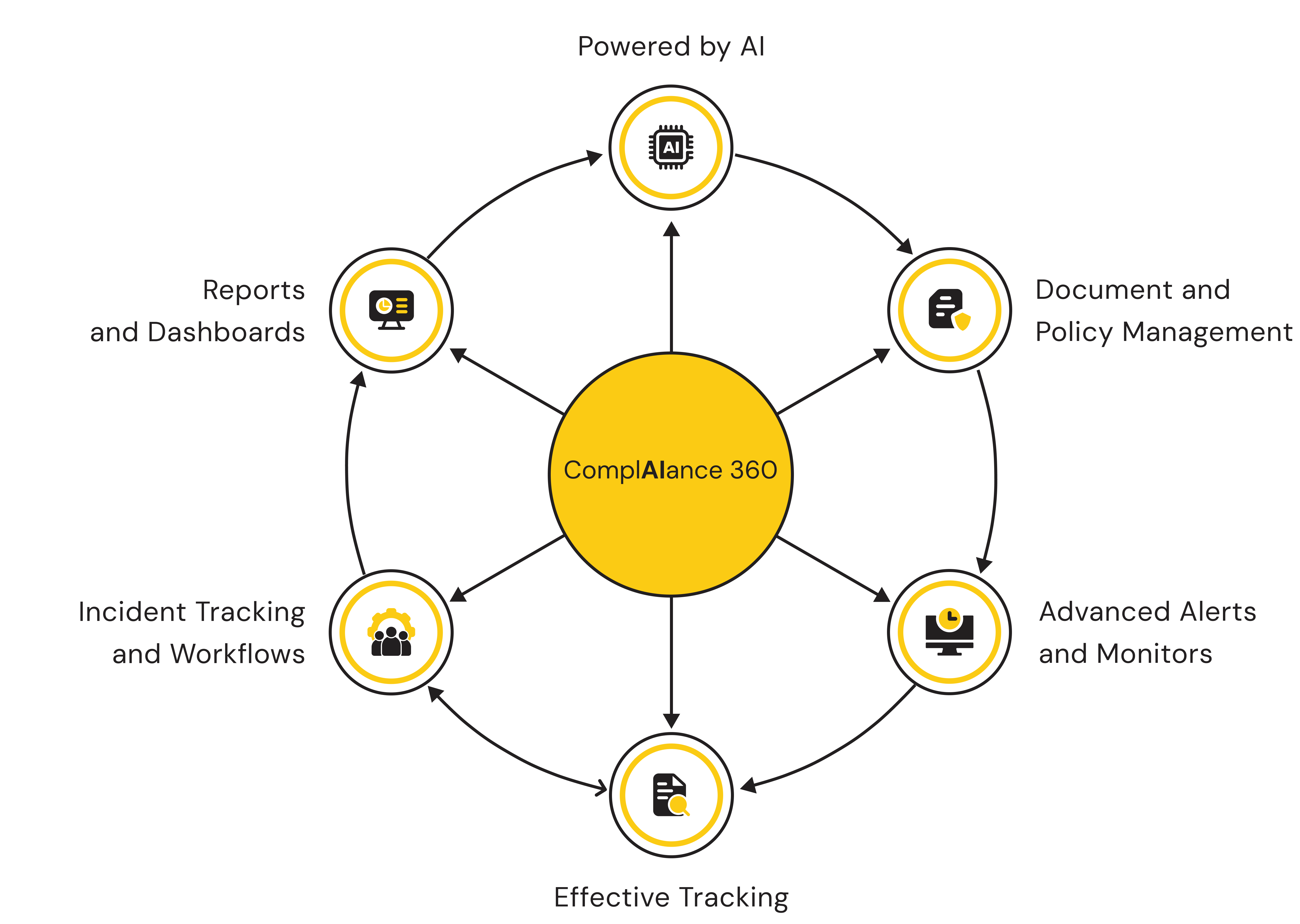
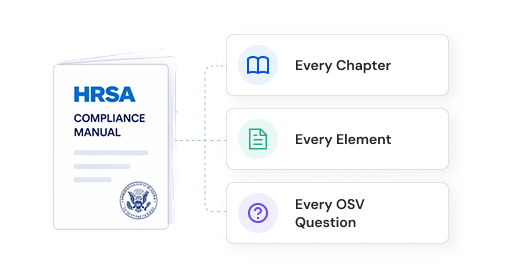
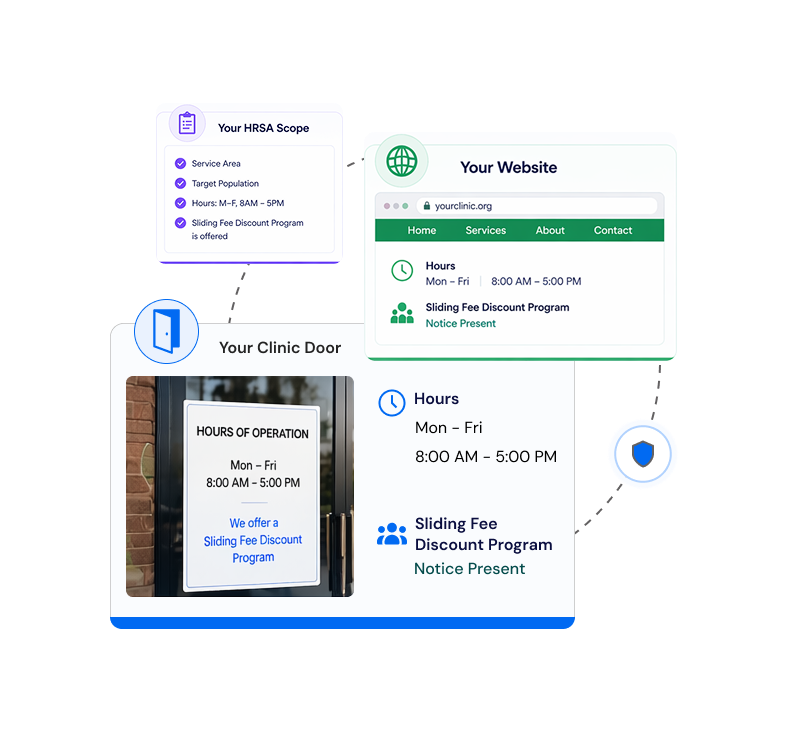
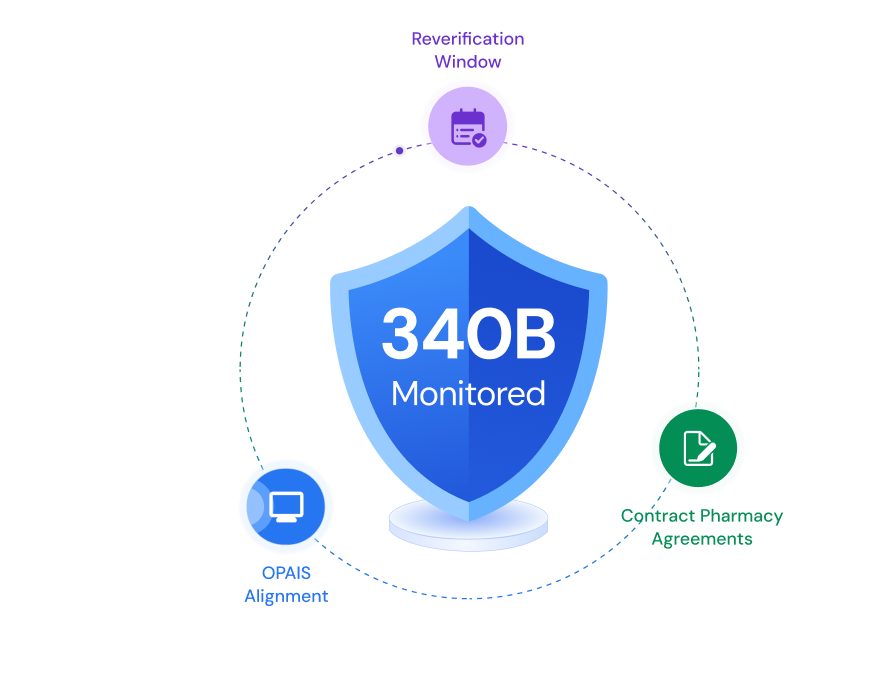
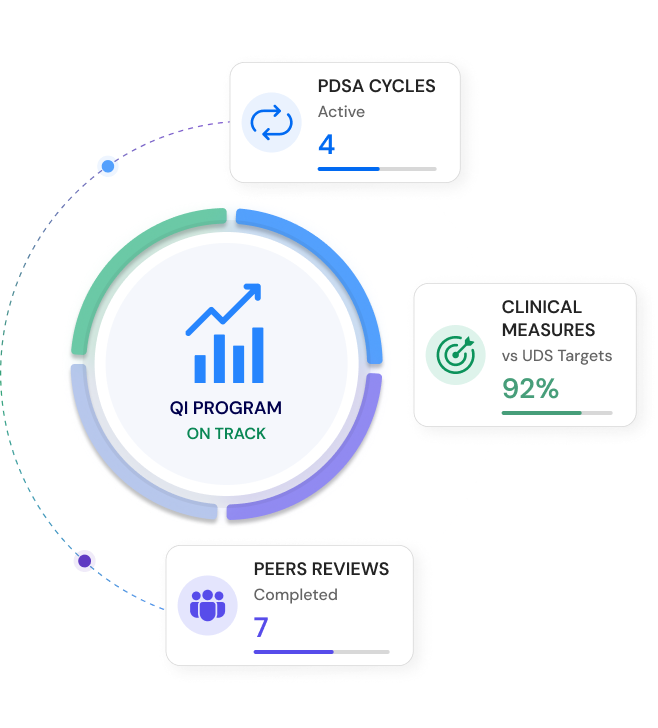
Compliance need not be cumbersome.
ComplAiance360 makes sure of it. Not a yearly scramble. A simplified, reliable compliance program that runs continuously — always on, always current, always ready when you need it.
Compliance should be part of the lifecycle, not an afterthought.